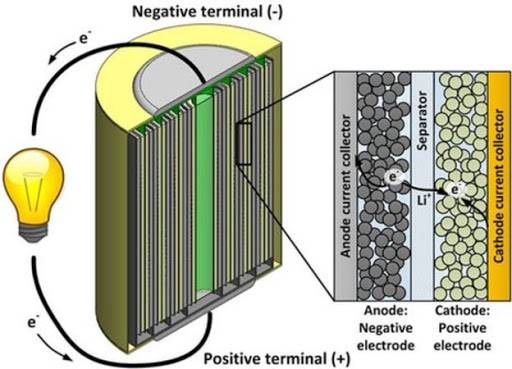
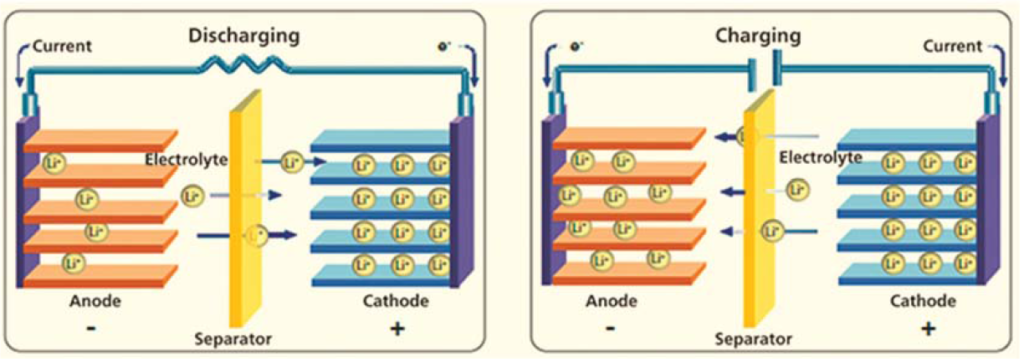
Lithium-ion batteries (LIBs) are a family of rechargeable batteries having high energy density and commonly used in consumer electronics. When a LIB is discharging, lithium ion move from the negative electrode (anode) to the positive electrode (cathode). When a LIB is charging, lithium ions move in the opposite direction. In general, the cathode material is composed of Iron-base, Cobalt-base, Nickel-base or Manganese-base transition metal oxides and the anode material is composed of graphite material. Both cathode and anode are manufactured using a layered structure and the lithium ions are located between layers (see schema below).
WHY LI-ION BATTERIES ARE THE FUTURE ?
Below are some of the reasons Lithium-ion batteries have found their way into almost all types of equipment so far
High specific energy
Lithium-ion batteries offer greater specific energy versus lead-acid (<40% weight compared to lead-acid) and other types of rechargeable batteries which enables batteries to become smaller and lighter.
Longer run time
This allows multi-shift crews to use Lithium-ion batteries power for longer periods of time over the course of a day and charge the battery when convenient
High charging efficiency
The differences with Lithium-ion batteries lie in a higher voltage per cell, tighter voltage… Charge efficiency is about 99% and the cell remains cool during charge.
Low capacity loss at high discharge rates
Compared to other rechargeable batteries, Lithium-ion batteries have a low capacity loss at high discharge rates and very good cycle life, as well as simple to charge and maintain.
Maintenance free
Lithium-ion is a low maintenance battery, an advantage that most other chemistries cannot claim.
Fast charging
Lithium-ion batteries can be designed for a fast charge of standard <3 hours.
Pollution free
Lithium-ion batteries do not contain mercury, cadmium or lead.
High energy density
Lithium-ion batteries are operate over a wider temperature range with higher energy densities than technologies such as NiCd, lead-acid or Ni-MH.





Thanks for your blog, nice to read. Do not stop.